Successful carbon nanotube production is dependent on a decent rate of growth and a process that can precisely create the nanomaterial required. At present, long production times and uncertainty over the quality of product and the specific properties of the carbon nanotubes grown is a major hurdle against the expansion of the nanomaterial industry.
Now a team, led by Feng Ding of the Center for Multidimensional Carbon Materials within the Institute of Basic Science in South Korea, have found a new process that can create specified types of carbon nanotubes (CNTs) with a selectivity of 90%. A process that offers CNT producers a great deal of promise for more accurate nanotube production.
Due to their strength, low weight, and high thermal and electrical conductivity, carbon nanotubes are highly sought-after raw materials, especially for electronic applications. However, accurate control over their synthesis can make nanotube production a ‘hit and miss’ process.
Carbon nanotubes are often imagined as a sheet of graphite just one atom thick (graphene) that has been rolled up into a tube. The properties of the tube depend on its length, diameter, and its chirality (the structure and way that individual atoms are connected).
If the graphene is rolled up on its long side, the tube will have different properties to if the graphene was rolled up on its short side or diagonally.
In this way, carbon nanotubes can be made as highly electrically conductive, or not conductive at all, or partially conductive. They can be made very strong, or very flexible, or neither strong nor flexible.
However, carbon nanotubes are not made by rolling up graphene, they are synthesized by adding carbon atoms one on top of another, on top of a rim. This slowly forms a tube, one nanometre at a time.
Despite the first nanoscale objects being created thirty years ago, our understanding of carbon nanotube synthesis is still quite limited, and nanomaterial producers struggle to make specific products accurately. However, accuracy is vital for a successful nanotube industry that relies on nanoproducts as high-quality raw materials for items such as energy efficient computer chips.
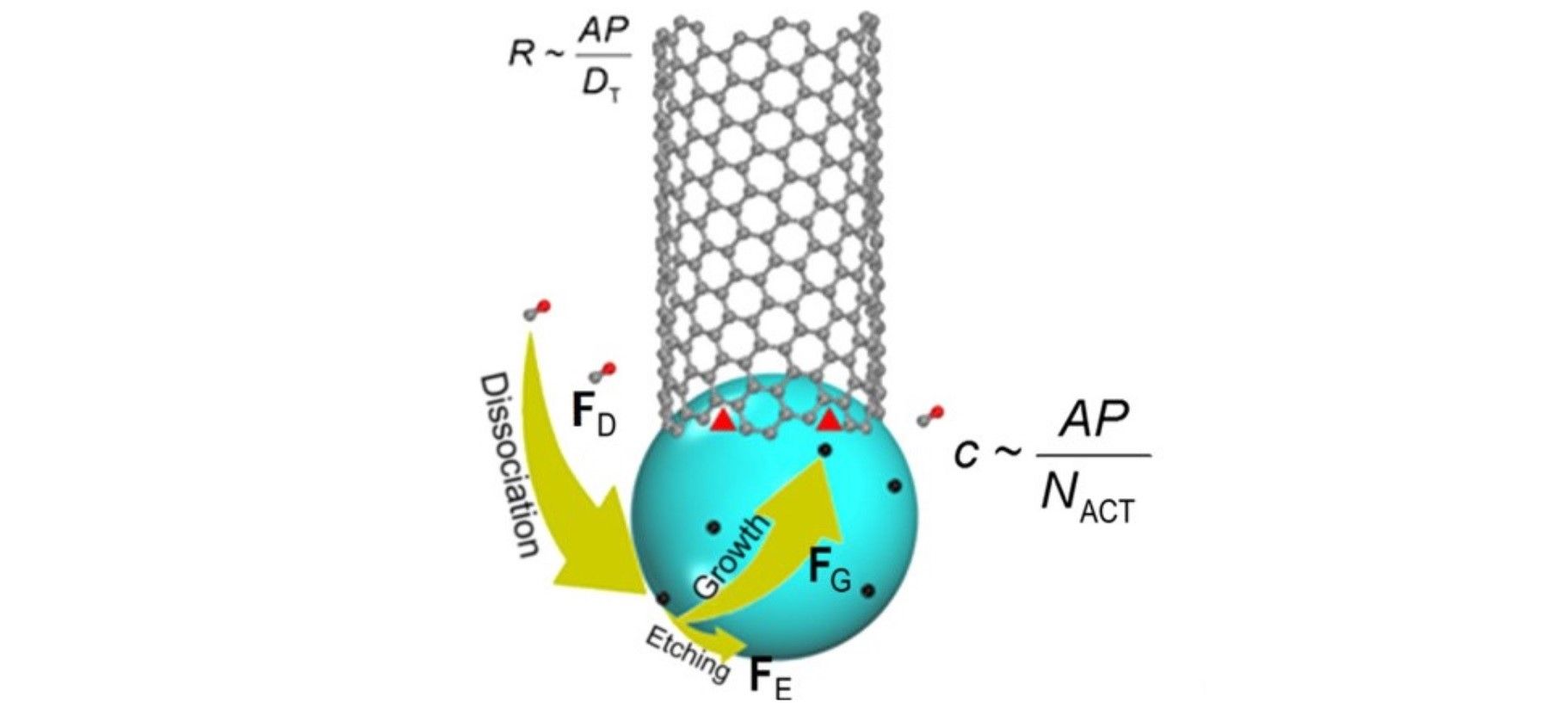
To date, one of the most promising manufacturing processes for CNTs is via chemical vapor deposition (CVD). This process combines metal nanoparticles (acting as a catalyst) and carbon-containing gases in a furnace. The metal nanoparticles dissociate the carbon from the gas, depositing them on the tube walls at their tip, making the tubes longer and longer, atom by atom. The growing stops when the catalyst particle is ‘encapsulated by graphitic or amorphous carbon’.
The growth rate is proportional to the density of the active sites available at the point where the CNT and catalyst meets.
However, in experiments that analysed the growth of CNTs on a magnesium oxide (MgO) support in a furnace at 700oC with carbon monoxide (CO) as a carbon feedstock and using cobalt nanoparticles as a catalyst, the Korean team found something different.
As Maoshuai He, the study’s first author, observes, “It was surprising that the growth rate of carbon nanotubes only depends on the size of the catalyst particle. This implies that our previous understanding on carbon nanotubes growth was not complete.”
Specifically, “… carbon atoms that are deposited on the catalyst particle surface can be either incorporated on the active side of the CNT or removed by etching agents, such as H2, H2O, O2, or CO2,” explains the online journal Science Daily. As a result, the team found that CNT growth rate depended on the ration between tube diameter and catalyst surface area.

The discovery could lead to, “… a new mechanism to selectively grow a specific type of CNTs, denoted as (2n, n) CNTs, which is characterized by the maximum number of active sites at the interface between the CNT and the catalyst. This CNT structure would correspond to rolling a sheet of graphite diagonally at an angle of around 19 degrees.”
“Compared to the previous model, we added three more factors: the rate of precursor deposition, the rate of carbon removal by etching agents, and the rate of carbon insertion into a carbon nanotube wall,” explains Ding, a leading researcher at the Center for Multidimensional Carbon Materials. “When feedstock dissociation cannot be balanced by carbon etching, the rate of carbon nanotube growth will no longer depend on the structure of the carbon nanotube. On the other hand, the previous theory is still valid if the etching is dominating.”
With this new understanding, the team were able to synthesize (2n, n) carbon nanotubes with selectivity of up to 90%. Achieving the highest selective growth with a high feedstock concentration and by excluding etching agents.
“If there is no carbon etching and the carbon nanotubes growth is slow, carbon atoms on the catalyst surface will accumulate," says Jin Zhang, a professor at Beijing University, and a co-author of the study. “This may lead to the formation of graphitic or amorphous carbon, which are established mechanisms of carbon nanotube growth termination. In this case, only carbon nanotubes which are able to add carbon atoms on their walls, that is with the highest number of active sites, can survive.”

The researchers have now published their findings in the journal Science Advances, where they, “… propose a more comprehensive model for SWCNT growth kinetics, where the carbon concentration on a solid catalyst surface is balanced by the rate of precursor deposition, the rate of elimination by etching agents, and the rate of carbon incorporation into the active sites of the SWCNT.”
While the specifics of the breakthrough are highly technical, nanomaterial industry workers are excited at the prospect of a new process to produce nanotubes with greater precision. It is also thought that an improved understanding of how the tubes grow will help manufacturers to speed up production, and consequently lower nanotube prices.
At present, the high production cost of nanotubes, coupled with a lack of certainty over quality are major drawbacks to the emergence of the nanomaterial industry. With further study continuing to improve nanomaterial production and the range of applications for these tiny tubes, this discovery could have major repercussions for raw material suppliers everywhere.
Photo credit: Cheaptubes, Science Advances, & Discovermagazine
